M Cell Autologous Fibroblasts
I. Patented Technology Advantages: High-Efficiency, High-Activity M Cell
M Cell utilizes autologous fibroblasts derived from an individual’s post-auricular (behind-the-ear) skin. Through MetaTech’s exclusive patented technology and precisely controlled culture conditions, M Cell demonstrates significantly enhanced performance compared with conventional single-cell products—achieving an eightfold increase in growth rate, a threefold increase in collagen secretion, and a threefold enhancement in cellular activity.
Beyond improving the cells’ inherent repair and regenerative potential, M Cell rapidly adheres to and integrates into damaged tissues after administration, enabling more predictable and long-lasting therapeutic outcomes.

__25J02d5T6q.png)
II. PIC/S GMP–Grade Cell Manufacturing: Three Pillars of Quality, Safety, and Consistency
M Cell is manufactured entirely within an internationally compliant PIC/S GMP–certified cell manufacturing facility. From tissue collection and cell isolation to activation, expansion, and final formulation, every step is conducted under strict aseptic conditions and comprehensive quality monitoring systems.
Each production batch undergoes multiple critical quality control tests, including cell viability assessments and sterility testing, to ensure that M Cell consistently delivers high safety standards and stable performance. This rigorous manufacturing control not only enhances product consistency but also reduces clinical implementation risks, positioning M Cell as a cell therapy product approved by the Ministry of Health and Welfare.
III. Core Differences from Conventional Products: From “Filling” to True “Regeneration”
Compared with traditional foreign-material fillers—such as hyaluronic acid—which primarily stimulate tissue response or provide temporary structural support, M Cell, as a cell therapy product, directly replenishes the skin’s most critical structural cells: autologous fibroblasts. Through the activity of highly viable cells, M Cell rebuilds the collagen and elastin framework, addressing skin aging at its root cause.
As an autologous cell–based therapy, M Cell offers high biocompatibility, significantly reducing the risk of foreign-body reactions while enhancing overall treatment safety. This cell-driven biological regenerative mechanism delivers more natural, long-lasting, and sustainable improvements in skin quality. Clinical evidence demonstrates outstanding outcomes in improving wrinkles, atrophic scars, acne scars, skin laxity, and other structural skin defects.

The images shown are for individual case reference only. Actual results may vary depending on individual physiology and skin condition. Treatment planning should be determined by a qualified physician following professional evaluation.
IV. Why Choose MetaTech Biomed’s M Cell Technology?
When selecting a cell therapy, safety, regulatory compliance, and technical expertise are critical. MetaTech Biomed’s M Cell technology is supported by multiple levels of clinical validation and regulatory approval—making it a choice you can trust.
1. Therapy Approved Under the Special Regulation for Cell Therapy
M Cell is an autologous fibroblast transplantation therapy approved by Taiwan’s Ministry of Health and Welfare under the “Special Regulation for Cell Therapy.”
It is clinically authorized for the treatment of wrinkles, depressions, scars, and other skin defects, positioning MetaTech Biomed as one of the few qualified cell preparation institutions in Taiwan with approved clinical implementation capability.
2. PIC/S GMP Manufacturing Ensuring Stable and Controllable Quality
MetaTech operates Taiwan’s first PIC/S GMP–certified cell manufacturing facility that meets international standards.
Each M Cell product undergoes approximately 45 days of rigorous manufacturing and quality testing, ensuring cell purity, safety, and consistent therapeutic performance.
3. Patents and National-Level Recognition
The M Cell technology has been granted two Taiwan invention patents and honored with the 16th National Innovation Award, demonstrating its technological leadership in the field of regenerative medicine.
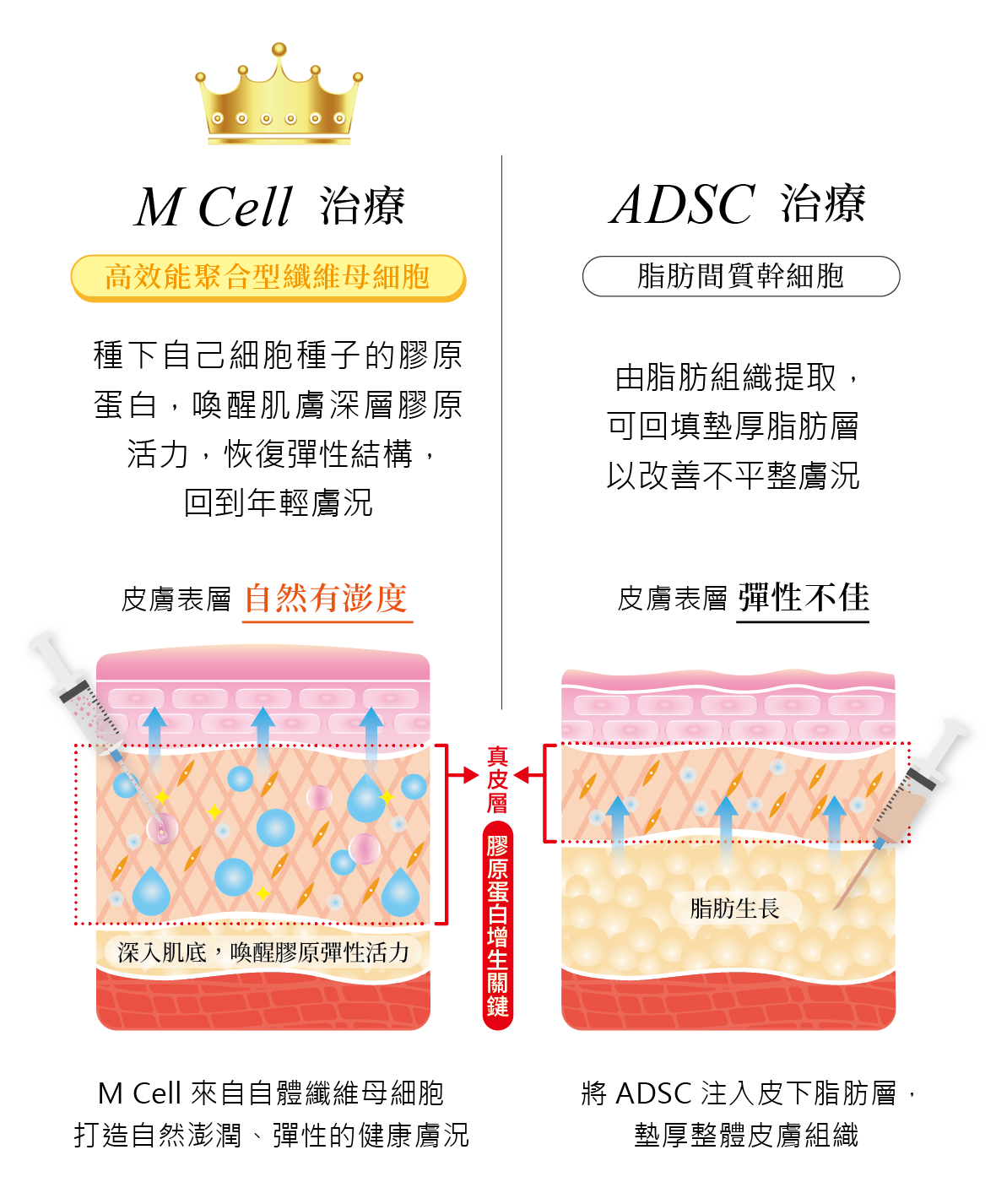
How Does M Cell Therapy Differ from Adipose-Derived Stem Cell (ADSC) Therapy?