From System to Execution
From System to Execution, Precisely Implemented Every Day
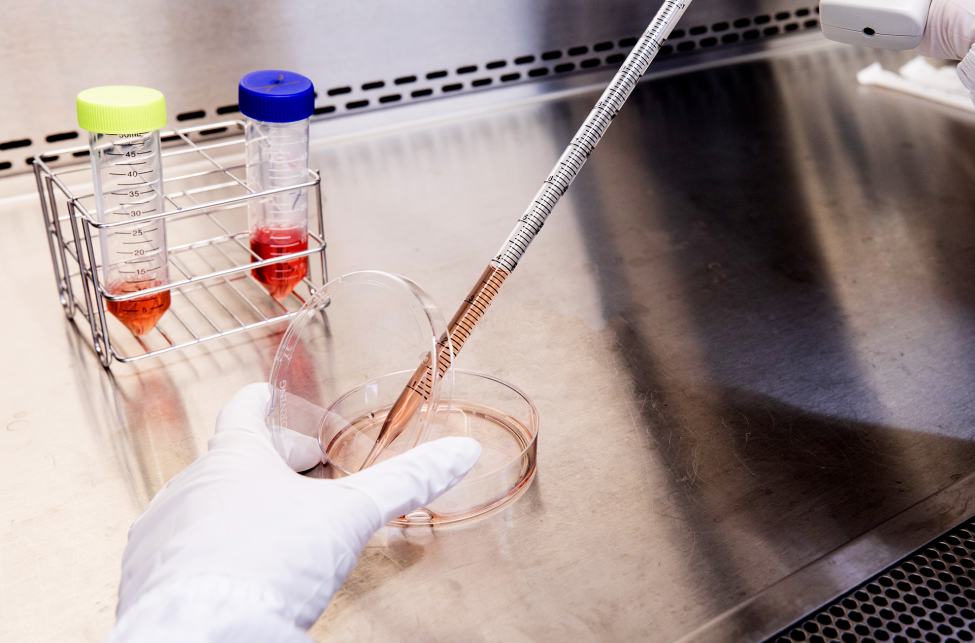
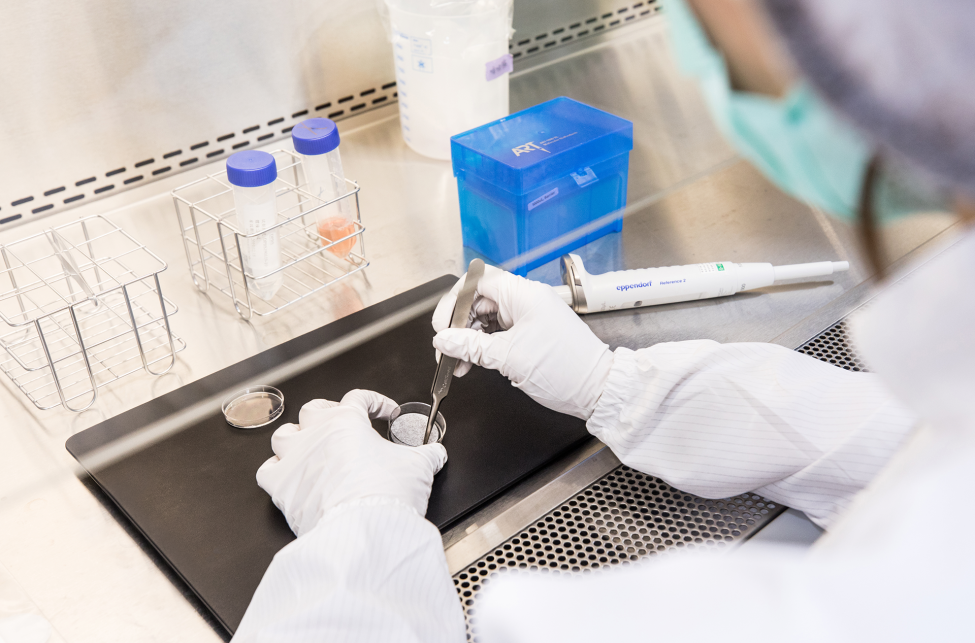
Cell manufacturing is a process characterized by high individual variability and low tolerance for error. Transforming it into a product that can be trusted depends on the ability to do things right every day and maintain consistent, stable quality operations.
This is why we chose to challenge ourselves with the PIC/S GMP certification, one of the most stringent international standards. It is not merely a qualification threshold, but a comprehensive test of operational rigor, quality management systems, and execution discipline.
From cleanroom design and automated monitoring to process documentation and personnel training, we have invested years in building a manufacturing system that is verifiable, sustainable, and consistently executable.
Making High Standards Actionable—and Scalable
-High standards are not only meant to be met, but to endure and expand.
-
2023
We built Taiwan’s first PIC/S GMP–certified cell manufacturing facility approved by the Ministry of Health and Welfare. -
2025
The successful renewal of our manufacturing license demonstrates the stability of our systems and our capability for sustained operations.
Beyond establishing robust systems and governance, we have further expanded our manufacturing capacity.
Behind every batch stands a team dedicated to quality.
Behind this rigorously safeguarded manufacturing system lies the long-term dedication and daily commitment of a team of professionals:
“In the beginning, the pressure was intense. We were afraid of making mistakes—because a single misstep at any stage could compromise the entire product.”
“Spending day after day in the cleanroom, repeating the same operations, can feel isolating. But we know exactly what we are holding ourselves to.”
“This is not just a task—it is a new way of working. A system that can be trusted, and one that can be executed consistently over time.”