MetaBiotech
Leading the Future of Cellular Medicine
MetaBiotech
Leading the Future of Cellular Medicine
THE POWER OF iPSC
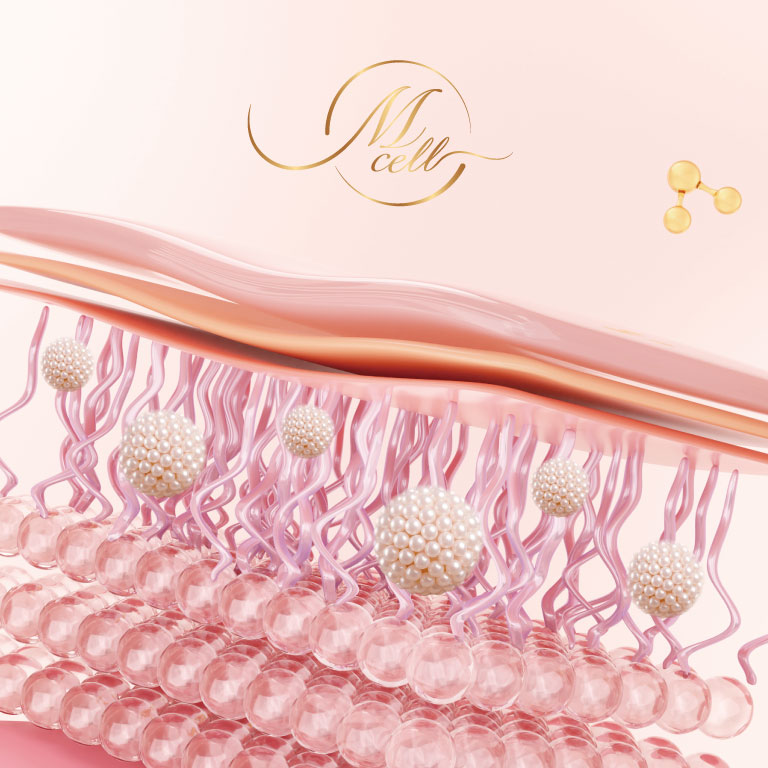
MetaTech Group is dedicated to the regenerative medicine industry, with a strong focus on two core technologies: induced pluripotent stem cells (iPSCs) and the patented M Cell technology. The Group continues to expand diverse applications in cellular technology, including exosomes, iPSC-EVs, S-PRP therapy, and the Exolimit brand, with a strategic emphasis on developing personalized regenerative medicine products.
Its subsidiary, Locus Cell Co., Ltd., has obtained global licensing from iPS Academia Japan, enabling the establishment of a comprehensive iPSC storage, manufacturing, and application platform. Through this integrated system, the company is driving the next era of precision regenerative medicine.
M Cell has been approved under the “Regulation Governing the Application of Specific Medical Examination Technique and Medical Device,” offering improved performance compared with traditional approaches. Developed using PIC/GMP clinical-standard manufacturing processes, it ensures safety and efficacy.
MetaTech Group collaborates with medical centers and research institutions worldwide, adhering to international GMP standards to become a key driving force in precision regenerative health.
__25L091jlLo.jpeg)


MetaTech Group
Innovation in Motion, the Foundation of Regeneration
MetaTech Group
Innovation in Motion, the Foundation of Regeneration
TOMORROW'S HEALTH STARTS HERE
Founded in 1998, MetaTech Group is an innovative biotechnology company based in Taiwan.
The Group operates Taiwan’s first PIC/S GMP–certified cell manufacturing facility approved by the TFDA, providing internationally compliant cell processing and quality control systems.
In recent years, MetaTech Group has further established Asia’s largest CDMO cell manufacturing facility in Zhubei City, integrating AI technologies and automated equipment to significantly enhance production efficiency and quality consistency. Through these advancements, the Group is building a solid manufacturing foundation for the regenerative medicine industry.
Powered by Core Strengths, Leading Regenerative Medicine
Powered by Core Strengths, Leading Regenerative Medicine
FUTURE BEGINS WITH CELLS
Our PIC/S GMP–compliant one-stop CDMO services encompass Nobel Prize–winning iPSC technologies, exosomes, iPSC-EV, autologous iPSC-EV, and autologous fibroblast manufacturing platforms—supporting both advanced R&D and scalable commercial production.
Globally Recognized GMP Quality Assurance
Professional R&D and technical team
CDMO Services
Intelligent regenerative medicine production model
Globally Recognized GMP Quality Assurance
Professional R&D and technical team
CDMO Services
Intelligent regenerative medicine production model







LATEST NEWS
LATEST NEWS
MEDIA REPORTS
MEDIA REPORTS
- Research & Development
MetaTech and CM Plus Establish Strategic Partnership to Build “AI Katana” Platform for Biotech Market Expansion in Japan and Asia
TOKYO/TAIPEI - October 13, 2025 - MetaTech (AP) Inc. (TWSE: 3224), a leading Taiwanese biotech AI technology company, and CM Plus, a multinational enterprise specializing in high-standard engineering management and regulatory compliance (headquartered in Singapore and Yokohama, Japan), have signed a Memorandum of Understanding (MOU) to jointly build a new cross-border platform for accelerating biotech product market entry between Taiwan and Japan.